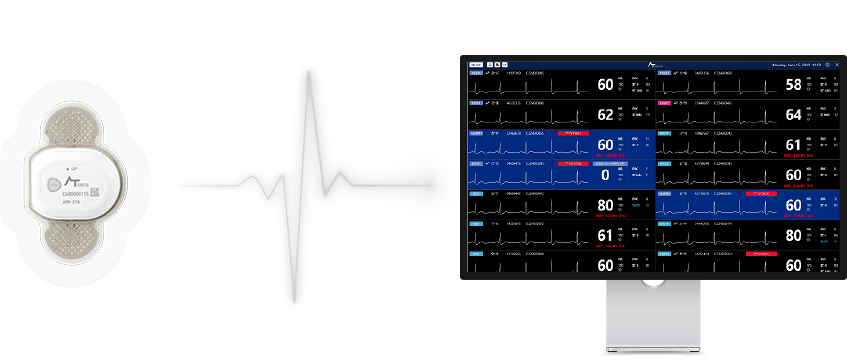
- AT-Patch
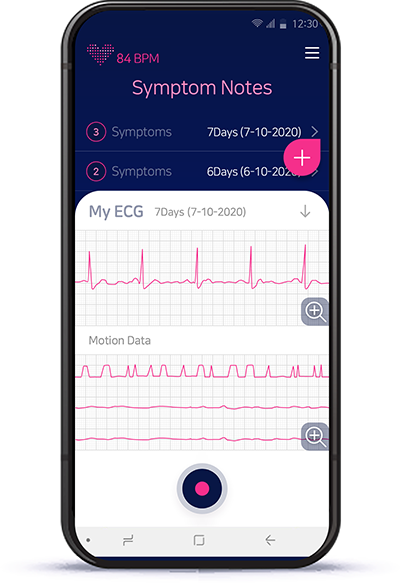
- AT-Note
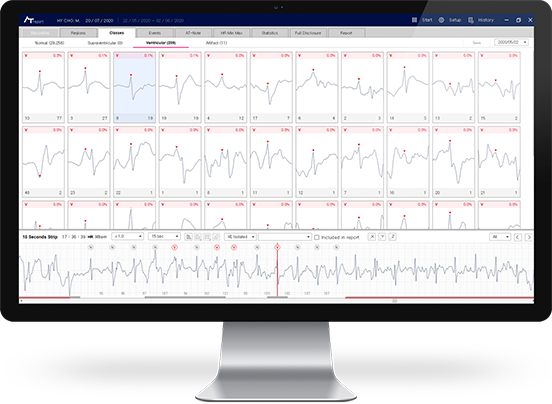
- AT-Report
- AT-Monitoring
AT-Patch's high accuracy detection
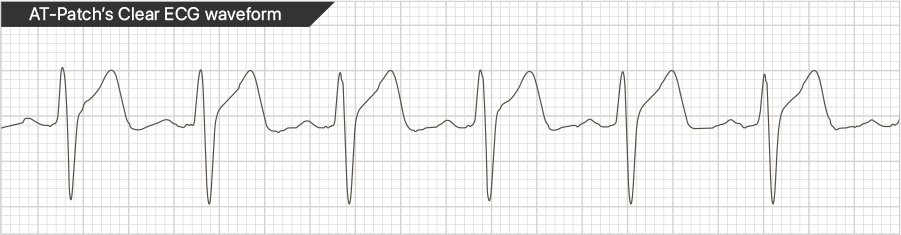 * ECG waveforms may have individual differences.
* ECG waveforms may have individual differences.  ATsens' advanced technology makes it possible to enhance high accuracy ECG detection. The sensor chip exclusively adopted to AT-Patch shows high performance to gather ECG signals from the heart and the short-length electrode built into the patch prominently reduces the inflow of noise(muscle signal) caused by body movement.
ATsens' advanced technology makes it possible to enhance high accuracy ECG detection. The sensor chip exclusively adopted to AT-Patch shows high performance to gather ECG signals from the heart and the short-length electrode built into the patch prominently reduces the inflow of noise(muscle signal) caused by body movement.
 Allows for showering and light activities
AT-Patch provides shower proof function. It meets the requirement of IP44 or IP57 grade so that it can be exposed to spray water. This function designed for patients helps to improve the quality of patient compliance during wearing it. Surely, it operates in the shower. It also operates while you work, walk, light activities, eat and sleep.
Allows for showering and light activities
AT-Patch provides shower proof function. It meets the requirement of IP44 or IP57 grade so that it can be exposed to spray water. This function designed for patients helps to improve the quality of patient compliance during wearing it. Surely, it operates in the shower. It also operates while you work, walk, light activities, eat and sleep.
 No charging or electrode replacement required while the ECG monitoring In the clinical field, long-time ECG monitoring is necessary to have a chance to find arrhythmias or irregular heartbeats that appear intermittently and get timely treatments. AT-Patch operates for up to 14 days(active mode) with only one single-use coin cell battery. Its high power consumption efficiency makes uninterrupted long-term ECG monitoring possible and makes maintenance-free.
No charging or electrode replacement required while the ECG monitoring In the clinical field, long-time ECG monitoring is necessary to have a chance to find arrhythmias or irregular heartbeats that appear intermittently and get timely treatments. AT-Patch operates for up to 14 days(active mode) with only one single-use coin cell battery. Its high power consumption efficiency makes uninterrupted long-term ECG monitoring possible and makes maintenance-free.
 Small size & light weight ATsens pitched in its technology to make the device optimized for a wearable and ambulatory ECG monitor. AT-Patch is compact size and only 13g weight. This patch-type device provides more comfortable experience.
Small size & light weight ATsens pitched in its technology to make the device optimized for a wearable and ambulatory ECG monitor. AT-Patch is compact size and only 13g weight. This patch-type device provides more comfortable experience. ECG Monitoring duration selectable
AT-Patch is the medical device optimized for long-term continuous ECG monitoring. It consists of a 7-day or 14-day model depending on the duration of the ECG monitoring.
ECG Monitoring duration selectable
AT-Patch is the medical device optimized for long-term continuous ECG monitoring. It consists of a 7-day or 14-day model depending on the duration of the ECG monitoring.
 Patient Monitoring AT-Monitoring
Patient Monitoring AT-MonitoringATP-T70 Real-time ECG Monitoring of patients admitted to a ward with wearable ECG Monitor ‘AT-Patch’.
It transmits the patient’s ECG signal to check their health.
Specification
| Model | ATP-C70 | ATP-C130 |
|---|---|---|
| Classification | Electrocardiographic Holter Analysis | |
| Monitoring Duration | Up to 7 days | Up to 14 days |
| Type | Disposable, Single lead (Lead Ⅱ) | |
| Medical device Classification |
Class Ⅱa | |
| size | Body 32.6x39.6x7.7mm
Patch 74x47.1x8.4mm |
Body 31x39x7.8mm
Patch 84x48.8x8.5mm |
| weight | 13g | 13g |
| Ingress Protection | IP44 | IP57 |
| Certificate | PMDA, MFDS | FDA(Previous Model Only) CE, PMDA, MFDS |
| Memory | Internal Memory | |
| OS | Android, iOS | |
| Power / Voltage | Internal power supply, DC 3V (Li/MnO2, Type CR2032) | |
 If the downloaded file does not open, check the viewer program.
If the downloaded file does not open, check the viewer program.
 Download PDF Viewer
Download PDF Viewer
* Disclaimer
1. The above information is based on the products, ATP-C130 obtained FDA, CE MDD(Medical Devices Directive), PMDA in Japan, MFDS in South Korea and ATP-C70 obtained PMDA in Japan, MFDS in South Korea. Thus, the information is specifically for South Korea & Europe and can differ with countries.
2. AT-Note(App) is usable only if you own a compatible smartphone or tablet. And the usage time of AT-Patch may be affected, depending on the individual communication environment for AT-Note’s Live ECG. For more information, please refer to the user manual.
3. AT-Report(PC S/W) is software for medical institutions, not provided patients directly. Patients can review the analysis result with or from your doctor.
4. The above information can be updated repeatedly.